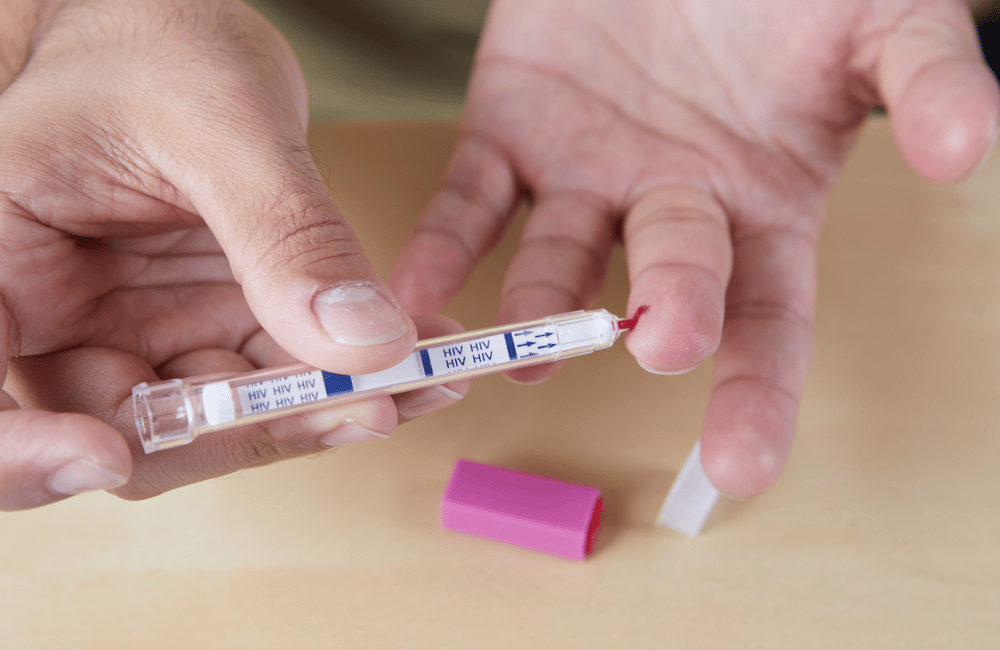
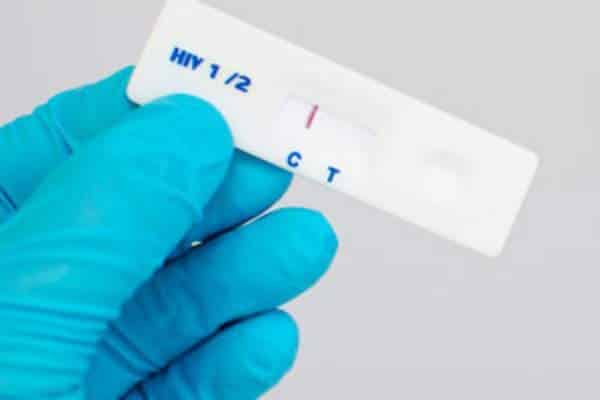
Galamsey activities fuelling HIV transmission in mining communities
Unregulated small-scale mining activities (galamsey) not only degrade the forests, pollute farmlands and river bodies with heavy metals and contribute to fatal health problems, but the activities also provide a conducive environment facilitating the spread of HIV at an alarming rate. According to research conducted by the Swiss Tropical and Public Health Institute, any time …