Description
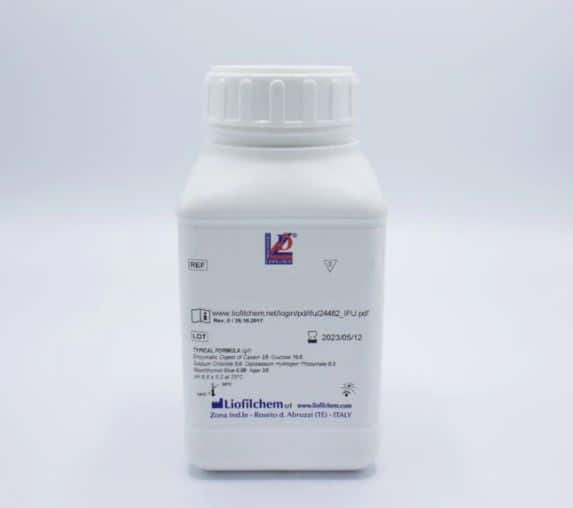
SIM Medium (From Liofilchem)
Semisolid medium for the identification of Enterobacteriaceae
by sulphide, indole and motility tests.
SIM Medium is used for the identification of microorganisms from clinical specimens and other samples on the
basis of hydrogen sulphide production, indole formation, and motility.
This medium is an aid to identify and discriminate between species of the family Enterobacteriaceae –
for example, Enterobacter has the general characteristics of Klebsiella species but can be differentiated because
they are motile as well as Citrobacter, Proteus, Providencia and Serratia; Klebsiella, Enterobacter, Hafnia and
Serratia species are usually indole negative whereas Escherichia species are positive for indole (except E.
vulneris); unlike Shigella, Salmonella species possess flagella and hence are motile and most produce hydrogen
sulphide (except S. Paratyphi A and S. Typhi, which is a weak producer).
Composition (g/litre)
Casein Peptone – 20.0
Meat Peptone – 6.1
Ferric Ammonium Sulphate – 0.2
Sodium Thiosulphate – 0.2
Agar – 3.5
Final pH – 7.3 ± 0.2 at 25°C
*Adjusted and/or supplemented as required to meet performance criteria.
METHOD PRINCIPLE
Peptones provide carbon, nitrogen and amino acids for bacterial growth. Casein peptone in particular contains
tryptophan which is converted to indole. Ferric ammonium sulphate and sodium thiosulphate are used to detect
hydrogen sulphide (H2S) production through formation of a black precipitate. Agar is the solidifying agent. The
low concentration of agar makes the medium semisolid allowing for visual determination of motility.
PREPARATION
Dehydrated medium: Suspend 30.0 g of the powder in 1 liter of distilled or deionized water. Mix well. Heat
to boil and shake until completely dissolved. Dispense into tubes. Sterilize in autoclave
at 121°C for 15 minutes.
Medium in bottles: Melt the content of the bottle in a water bath at 100°C (loosing the cap partially
removed) until completely dissolved. Then screw the cap and check the homogeneity
of the dissolved medium, if it is the case turning the bottle upside down. Cool at
45-50°C, mix well avoiding foam formation and aseptically distribute into tubes.
Note: Sufficient volume of medium must be dispensed into tubes to give a depth of about 4 cm. Allow tubes to
solidify in a vertical position.
TEST PROCEDURE
1. Inoculate test organism by stabbing two-thirds into the medium.
2. Incubate with loose caps at 35 ± 2°C for 18-24 hours.
3. After incubation, examine tubes for motility and H2S production.
4. Once H2S and motility have been recorded, add 3-4 drops of Kovac’s Reagent (ref. 87001) to each tube.
Note: Test organisms must be in pure culture. The inoculum should be taken from a solid medium as inoculum
from liquid suspensions may delay results. Erroneous results may occur if caps are not loose during incubation.
INTERPRETING RESULTS
Motility: A positive motility test is indicated by a diffuse growth/turbidity extending from inoculating
stab line, whereas non-motile organisms grow only along the line of inoculation.
H2S: Hydrogen sulphide production is shown by a blackening of the medium in those areas where
microbial growth has occurred.
Indole: Indole formation is seen as appearance of a pink or red color, whereas test is negative if there is
no color change after addition of Kovac’s Reagent.
Consult appropriate references for activities of specific microorganisms and for complete identification of
Enterobacteriaceae.
Weight: 500g
Related products
-


10mL Disposable Syringe
₵125.00 Add to cart -
 Sale!
Sale!

HemoCue Hb 301 Microcuvettes
₵1,550.00Original price was: ₵1,550.00.₵1,450.00Current price is: ₵1,450.00. Add to cart -


Gel Blood Collection Tube
₵105.00 Add to cart -

Gypsona POP
₵250.00 Add to cart -


5mL Disposable Syringe
₵115.00 Add to cart -

Urinalysis Reagent Strips, Urit 10V
₵170.00 Add to cart